For years, sickle cell disease had no cure. This month, the Breakthrough Prize, the world’s most prestigious science award given annually to recognise transformative advances in life sciences, physics, and mathematics, went to the researchers who finally changed that.
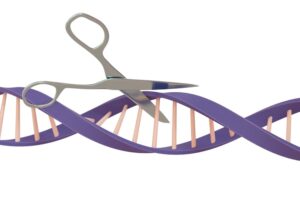
CRISPR, which stands for Clustered Regularly Interspaced Short Palindromic Repeats, is a revolutionary gene-editing technology that allows scientists to locate and correct errors in DNA, the genetic blueprint inside every cell of the human body, with extraordinary precision.
The therapy the Breakthrough Prize winners helped build, called Casgevy, is a marvel of modern medicine. It is also priced at approximately $2.2 million (roughly Rs 18 to 20 crore) per patient abroad, a number that puts it permanently out of reach for the communities in India who need it most.
In a laboratory in New Delhi, a team of Indian scientists has spent years quietly building an answer to that problem. They call it BIRSA 101. And if it clears human trials, it could change the rules of gene therapy for an entire country.
THE DISEASE THAT BREAKS BLOOD
In a healthy person, red blood cells are round, soft, and flexible. They glide through blood vessels, carrying oxygen to every organ.
In someone with sickle cell disease, a single typo in the genetic code, one wrong letter in a three-billion-letter instruction manual, causes these cells to twist into a crescent or sickle shape.
These rigid, misshapen cells clog blood vessels, starve organs of oxygen, and cause episodes of excruciating pain called crises.
Over years, they quietly damage the heart, lungs, and kidneys. Many patients do not survive past middle age.
India carries one of the heaviest burdens of this disease in the world. It is especially prevalent in the tribal belts of Jharkhand, Chhattisgarh, Madhya Pradesh, and Odisha, where access to basic medical care is often hours away.
THE SCISSORS THAT REWRITE LIFE
Sickle cell disease is caused by a single misprint in the body’s genetic instruction manual. CRISPR is a technology that can find that misprint and fix it.
It works using a protein called Cas9, which acts like molecular scissors. A protein is a biological molecule that performs specific tasks inside the body. A separate guide molecule, essentially a GPS signal, directs these scissors to the exact location of the error in the DNA.
The scissors cut, and the cell repairs itself using a corrected version of the instruction.

In gene therapy for sickle cell disease, doctors remove blood-forming stem cells from the patient’s bone marrow. Stem cells are master cells that can grow into any type of blood cell. The bone marrow is the spongy tissue inside bones where these cells are made. The stem cells are then sent to a laboratory where the genetic edit is made, and returned to the body.
If the corrected cells settle in, a process called engraftment, and multiply, they can produce healthy blood for the rest of the patient’s life.
A SHARPER PAIR OF SCISSORS
Casgevy uses a widely available protein called SpCas9**, sourced from a common bacterium.**
BIRSA 101 uses a custom-built alternative called enFnCas9, designed and engineered at CSIR-IGIB, the Council of Scientific and Industrial Research’s Institute of Genomics and Integrative Biology, in New Delhi.
Dr Souvik Maiti, one of the inventors of BIRSA 101 along with Debojyoti Chakraborty, told IndiaToday.in: “The biggest advantage of BIRSA 101 lies in precision and safety. The CRISPR system used in Casgevy relies on SpCas9, which is effective but can sometimes bind or cut at unintended sites in the genome.”
The genome is the complete set of DNA instructions inside a human cell, containing roughly three billion letters of genetic code.
He added: “Our engineered enzyme, enFnCas9, is designed to be far more accurate. It has an inherent ability to distinguish even a single mismatch in DNA and avoids binding to incorrect sites. In simple terms, it is like having a sharper and more intelligent pair of molecular scissors, which improves both safety and reliability.”
There is also a second important difference. Casgevy reactivates a backup gene that produces fetal haemoglobin, a type of haemoglobin the human body makes only before birth and then switches off. Haemoglobin is the protein in red blood cells responsible for carrying oxygen. BIRSA 101 goes straight to the source of the problem.
Dr Maiti explained why this matters for Indian patients. “While some therapies work by increasing fetal haemoglobin, that strategy may not be optimal for Indian patients, because many already have relatively higher levels of fetal haemoglobin. So further increasing it may not deliver a consistent benefit. By correcting the root genetic defect, we aim for a more definitive and universally effective solution.”
THE SAFETY NUMBERS THAT MATTER
The greatest fear surrounding any CRISPR therapy is off-target editing, where the molecular scissors accidentally cut the wrong part of the DNA, potentially triggering dangerous mutations elsewhere in the genome.
“We performed genome-wide editing studies,” Dr Maiti said, referring to tests that scan the entire DNA to check whether the editing protein attaches itself only where it should. “These showed far fewer unintended interactions compared to existing systems. In cell-based editing experiments, we observed no detectable off-target edits at known problematic sites.”

“Highly sensitive genome-wide assays, which are detailed laboratory tests that examine the entire genetic code for errors, showed up to 1,000-fold fewer off-target events compared to conventional CRISPR tools. Taken together, these results give us strong confidence that BIRSA 101 edits only the intended DNA site with very high precision,” he added.
A 1,000-fold improvement in precision is not a minor technical footnote. It is a meaningful leap in the safety profile of the technology.
A CURE WRITTEN IN STEM CELLS
Dr Maiti said the therapy targets blood-forming stem cells, which naturally regenerate the entire blood system.
“If these cells are successfully edited and engrafted well, they can continue producing healthy blood cells for life. So the goal is a one-time, potentially lifelong cure. However, as with any new therapy, long-term clinical follow-up is essential, and that evidence will come as trials progress,” he explained.
Phase 1 human trials will soon be initiated. Patient outcomes will be reported as the trial progresses.
THE PRICE OF A CURE
Casgevy is priced at approximately $2.2 million (roughly Rs 18 to 20 crore) per patient abroad. Reports estimate BIRSA 101 could eventually be available for around Rs 50 lakh, a fraction of that cost.
Dr Maiti said it is too early to confirm an exact price, but the therapy is expected to be dramatically more affordable, potentially 15 to 20 times lower than therapies priced abroad.
“This is due to the use of an indigenously developed CRISPR system avoiding expensive licencing, Indian manufacturing scale including partnerships like Serum Institute, and strong public-sector and government support. Our goal is to make this therapy accessible, not just available,” he added.
A cure that exists only in the bank accounts of the wealthy is not really a cure at all.
THE NAME THAT CARRIES A PROMISE
BIRSA 101 takes its name from Bhagwan Birsa Munda, the revered Adivasi leader and freedom fighter, on his 150th birth anniversary.
Sickle cell disease falls hardest on India’s tribal communities, the very communities Birsa Munda spent his life fighting for.
The challenge of delivering this therapy to remote tribal areas remains significant.

Dr Maiti envisions a hub-and-spoke model, where a central advanced hospital acts as the hub and smaller regional clinics act as spokes, bringing care closer to patients who cannot travel far.
“At present, we are focused on establishing safety and efficacy through clinical trials. Large-scale deployment plans will follow successful completion of Phase 1, 2 and 3 trials. The challenge is significant, but it is not insurmountable,” Dr Maiti said.
The Breakthrough Prize this year celebrated the scientists who proved gene therapy could work. BIRSA 101 is India’s attempt to prove it can work for everyone.