ast year, 11-year-old Aarav Mehta, from Ahmedabad, stopped responding to standard treatment for a rare blood disorder. Instead of another round of broad-spectrum drugs, doctors suggested something different: whole-genome sequencing, followed by a personalised therapy designed around the exact genetic mutation causing his disease. Within weeks, Aarav’s DNA was sequenced, the faulty gene pathway identified and he was put on a therapy designed for that mutation. A few months later, his symptoms stabilised. Cases like Aarav’s becoming the face of a new era in Indian healthcare—one where treatment is not based on a one-size-fits-all approach developed for the average patient, but on an individual’s DNA and unique disease profile.
L
“Instead of treating patients with the same disease in the same way, precision medicine tailors prevention and treatment based on individual differences in genes, environment and lifestyle,” says Dr Vikas Maurya, HOD, Department of Pulmonology & Sleep Disorders, Fortis Hospital, Delhi. “The idea is to move from population-based medicine to personalised medicine, where treatment decisions are guided by biological data.”
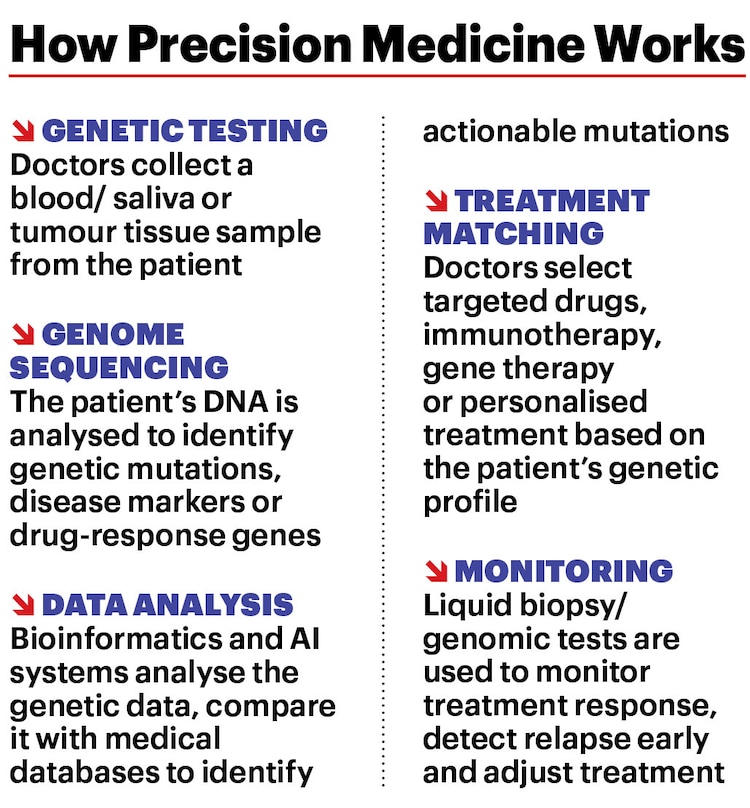
DNA (Deoxyribonucleic Acid) is a molecule made up of two strands of polynucleotides and holds genetic information inside cells, while genes are segments of DNA that contain instructions to build proteins, essential for our bodies to grow and function. They are the units of heredity. Genetic mutations are changes in the DNA sequence or genome—the linear arrangement of all gene sequences and other chemical bases—during cell division. If part of the DNA sequence is in the wrong place or incomplete, it might lead to genetic disorders and diseases. Genome sequencing, the laboratory process of determining the whole DNA sequence, can identify genetic causes of diseases.
“India has thousands of endogamous population groups with distinct genetic signatures,” says Arjun Gupta, founder & CEO, Genetico, a research and diagnostics firm. “This means population-specific genetic variants and disease risks are different from Western populations.” Government programmes like the Genome India Project have sequenced over 10,000 Indian genomes from 99 populations, creating a database meant to improve diagnosis, drug response prediction and treatment design. The Indian Cancer Genome Atlas is also trying to map mutation patterns specific to Indians.
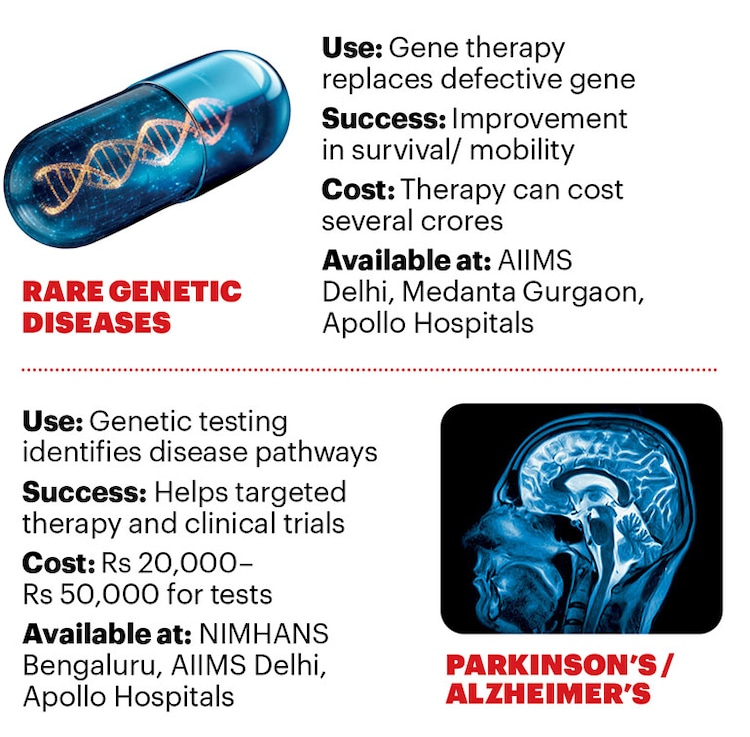
While precision medicine began in cancer care, its use is now expanding, especially in rare genetic disorders, cardiology and neurology. Indian hospitals are using genomic testing panels (tests that analyse multiple genes linked to cancer) for treatment selection and pharmacogenomic tests to identify medicines a patient is more likely to respond to. Gene-based treatments for conditions like spinal muscular atrophy and inherited retinal disorders are becoming available through specialised centres.
The developments can’t come soon enough. India accounts for 7–8 per cent of global cancer cases, nearly 27 per cent of the world’s tuberculosis burden and has an estimated 70 million people living with rare diseases, many of them genetic.
The Indian precision medicine market is expected to grow rapidly as genome sequencing becomes cheaper and more hospitals use genetic testing before prescribing treatment. Market research firm IMARC put the size of the market at $2.67 billion (Rs 24,898 crore) in 2025 and estimates annual growth at around 11–12 per cent.
THE NEW CANCER PLAYBOOK
Cancer remains the most fertile ground for precision medicine. In many cancers where decisions are based on tumour location and extent of malignancy, doctors can now identify specific mutations driving the disease.
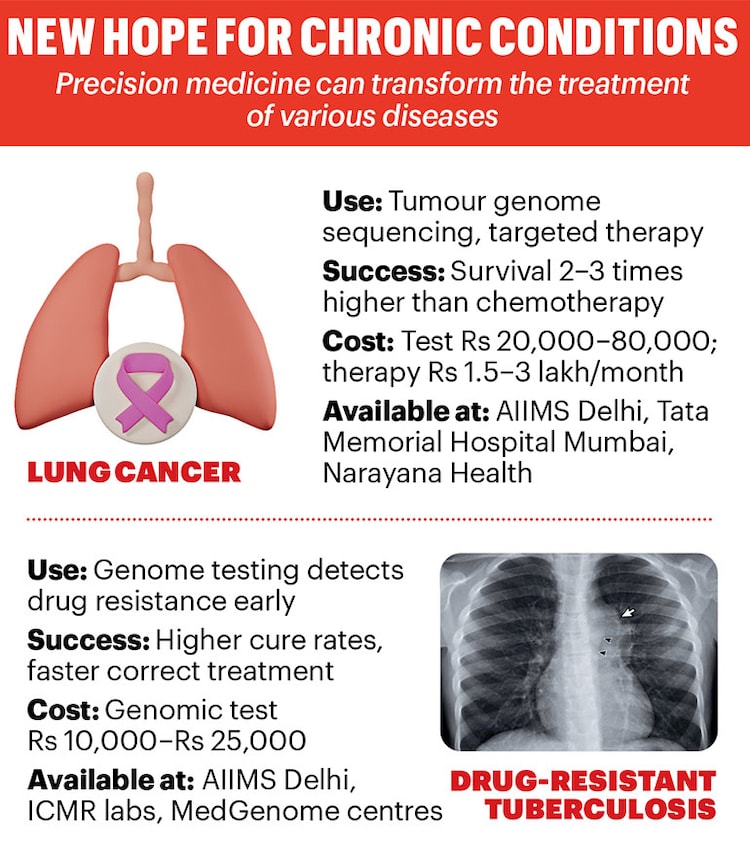
Rohit Menon, a 46-year-old chartered accountant from Kochi, was diagnosed with advanced non-small cell lung cancer. Instead of starting chemotherapy, his doctors performed tumour genome sequencing, which revealed an EGFR gene mutation. He took targeted therapy; within months, his tumour reduced and his disease stabilised. This is because targeted drugs block the mutation driving the cancer cells, rather than attacking all rapidly dividing cells the way chemotherapy does. For patients, this means fewer side-effects, longer disease control and a better quality of life.
A 2026 study in the Journal of Clinical Oncology reported that, in certain advanced cancers, patients whose treatment was guided by genome sequencing and matched targeted therapy had progression-free survival rates 30–40 per cent higher than those treated with conventional chemotherapy alone. Some cancers may be detected before symptoms appear. “Liquid biopsy tests can detect fragments of tumour DNA in blood, sometimes years before any symptoms, which could allow cancer to be treated earlier,” says Dr Maurya.
However, much depends on tumour biology and disease stage. “Tumour genome sequencing is increasingly being integrated into oncology practice in India, although its impact varies, depending on the type and stage of cancer,” says Dr Randeep Singh, director, Oncology Services, Narayana Hospital, Gurugram.
Genomic sequencing is influencing treatment in advanced lung cancer, bile duct cancer, advanced prostate cancer, breast cancer after multiple lines of therapy and metastatic colon cancer.
SMARTER MEDICINES
Besides cancer, precision medicine is increasingly being used to treat infectious diseases and rare genetic disorders. Tuberculosis, for one. Success in TB treatment depends on early detection. “Early detection allows clinicians to start the correct treatment earlier and prevent the spread of resistant strains,” says Dr Gunisha Pasricha, associate director, Infectious Disease Genomics at the genetic testing lab MedGenome. TB culture tests can take three–eight weeks to detect drug resistance. Molecular and genomic tests, such as GeneXpert/ CBNAAT, TrueNat and genome sequencing, can identify it within hours to a few days. That difference in time can change treatment outcomes. If needed, whole genome sequencing can detect mutations linked to resistance to 18 tuberculosis drugs, helping doctors to home in on the right medicine.
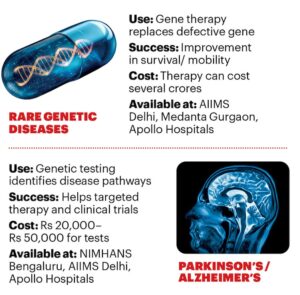
Thus, the main advantage of precision medicine is accuracy in treatment. Thanks to pharmacogenomics—the study of how an individual’s genetic makeup influences her body’s response to drugs—patients receive drugs that work for them. It reduces the trial-and-error approach that persists in treating depression, hypertension and diabetes. In cardiology in India, genetic tests for conditions such as familial hypercholesterolemia and inherited cardiomyopathies are now being used to identify high-risk individuals early. In neurology, genetic testing is increasingly used to diagnose and treat Alzheimer’s and Parkinson’s, epilepsy, spinal muscular atrophy and certain neurodegenerative disorders, where identifying the exact genetic mutation helps doctors choose targeted therapies.
Further, genomics also holds the promise of helping public health systems monitor disease trends, identify outbreak clusters earlier and track drug resistance in real time.
THE TALLEST HURDLES
Cost and infrastructure are the biggest barriers to speedier adoption of precision medicine in India. Many advanced genomic tests and targeted therapies are expensive and not covered by insurance. Tumour genome sequencing can cost from Rs 20,000 to Rs 80,000, while targeted cancer therapies can cost from Rs 1.5 lakh to Rs 4 lakh a month. Genome sequencing tests for drug-resistant tuberculosis can cost Rs 10,000 to Rs 25,000 in private facilities, limiting access outside government hospitals. Some gene therapies for rare diseases can cost several crores of rupees, making them inaccessible for most patients.
Cost is only part of the problem. Precision medicine requires specialised laboratories, bioinformatics systems and trained molecular scientists who can interpret genomic reports and guide treatment decisions. Many of these facilities are still concentrated in metropolises with large hospitals, like AIIMS Delhi, Tata Memorial Mumbai and the Narayana and Apollo hospital chains. “There are multiple barriers to implementing precision medicine in India,” says Dr Jyoti Wadhwa, principal lead of Medical and Precision Oncology, Apollo Athenaa Women’s Cancer Centre, Delhi. “Cost, infrastructure, lack of trained specialists and limited access to molecular tumour boards (panel of medical experts who review complex cancer cases based on genomics) are major barriers.”
Another major challenge lies in interpreting data generated from DNA sequencing alongside clinical data, treatment history and patient outcomes. In India, medical records are often unstructured and stored across multiple systems, making it difficult to combine genomic data with clinical information in a meaningful way. “The bigger barriers now lie in standardisation, interpretation of results and availability of trained specialists,” says Dr Singh. Genome sequencing produces massive amounts of information and these are increasingly processed through artificial intelligence and bioinformatics tools that turn complex results into simplified clinical reports.
The future of precision medicine in India lies not just in sequencing more genomes, but also in making that information usable. In the next big shift, genomic data will be integrated into everyday clinical decision-making. If that happens, precision medicine could truly transform diagnosis, treatment selection and disease prevention over the next decade. n